“High-risk early triple-negative breast cancer is frequently associated with early recurrence and high mortality. Neo-adjuvant chemotherapy is the preferred treatment approach. In addition to potentially increasing the likelihood of tumor resectability and breast conservation, patients who have a pathological complete response after neoadjuvant therapy have longer event-free survival (defined as the time from randomization to the date of disease progression that precluded definitive surgery, the date of local or distant recurrence or the occurrence of a second primary tumor, or the date of death from any
cause) and overall survival. Accordingly, regulatory guidance supports the use of the pathological complete response as an end point for clinical testing of neoadjuvant treatment in patients with early triple-negative breast cancer.”
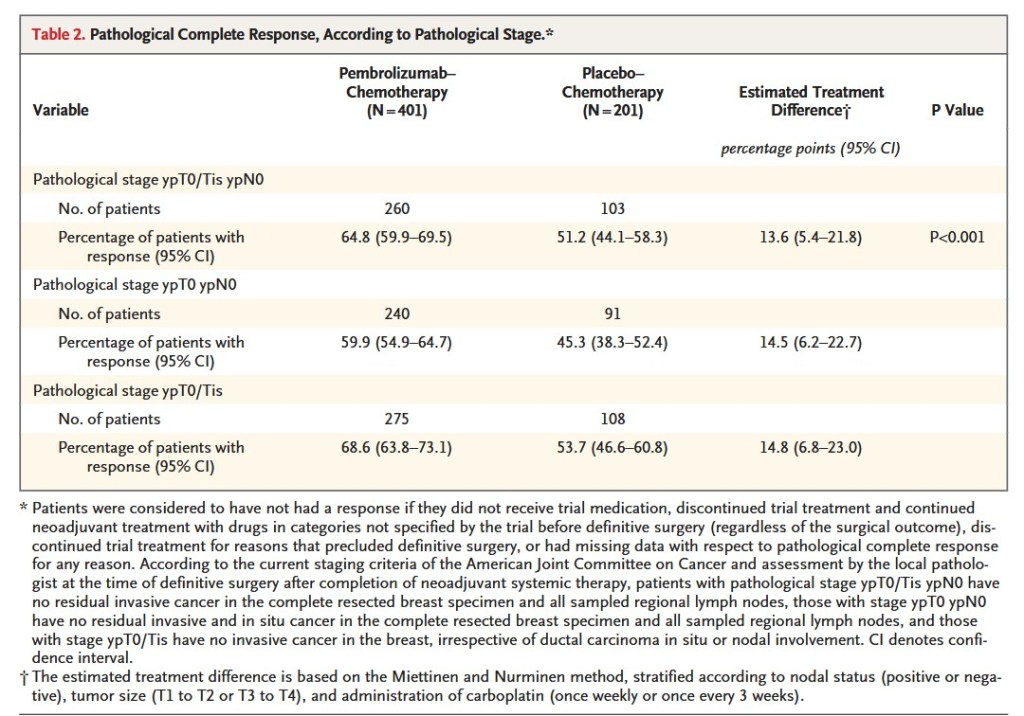
“In this randomized phase 3 trial involving patients with previously untreated, early triple-negative breast cancer, a significantly higher percentage of patients in the pembrolizumab–chemotherapy group than in the placebo–chemotherapy group had a pathological complete response at the time of definitive surgery. The benefit of pembrolizumab–chemotherapy with respect to pathological complete response was generally consistent across subgroups, including PD-L1–expression subgroups. This finding differs from the results of the IMpassion130 trial, which showed efficacy of a PD-L1 inhibitor only in patients with
PD-L1–positive metastatic triple-negative breast cancer; the inconsistent results may be related to the different drugs or inhibition pathways, disease stages (early rather than late), PD-L1 assays, or all of these factors. Analyses of molecular biomarkers that might predict a clinical response to pembrolizumab are ongoing in our trial. The percentage of patients with a pathological complete response in the placebo–chemo-therapy group was consistent with percentages reported in other studies of platinum-containing neoadjuvant regimens in patients with early breast cancer.”
Schmid, Peter et al. “Pembrolizumab for Early Triple-Negative Breast Cancer.” The New England journal of medicine vol. 382,9 (2020): 810-821. Full Text for Emory Users
