Maki DG, Kluger DM, Crnich CJ. The risk of bloodstream infection in adults with different intravascular devices: a systematic review of 200 published prospective studies. Mayo Clin Proc. 2006 Sep;81(9):1159-71. Full-text for Emory users.
Results: Point incidence rates of IVD-related BSI were lowest with peripheral Intravenous catheters (0.1%, 0.5 per 1000 IVD-days) and midline catheters (0.4%, 0.2 per 1000 catheter-days). Far higher rates were seen with short-term noncuffed and nonmedicated central venous catheters (CVCs) (4.4%, 2.7 per 1000 catheter-days). Arterial catheters used for hemodynamic monitoring (0.8%, 1.7 per 1000 catheter-days) and peripherally inserted central catheters used in hospitalized patients (2.4%, 2.1 per 1000 catheter-days) posed risks approaching those seen with short-term conventional CVCs used in the Intensive care unit. Surgically implanted long-term central venous devices–cuffed and tunneled catheters (22.5%, 1.6 per 1000 IVD-days) and central venous ports (3.6%, 0.1 per 1000 IVD-days)–appear to have high rates of Infection when risk Is expressed as BSIs per 100 IVDs but actually pose much lower risk when rates are expressed per 1000 IVD-days. The use of cuffed and tunneled dual lumen CVCs rather than noncuffed, nontunneled catheters for temporary hemodlalysis and novel preventive technologies, such as CVCs with anti-infective surfaces, was associated with considerably lower rates of catheter-related BSI.
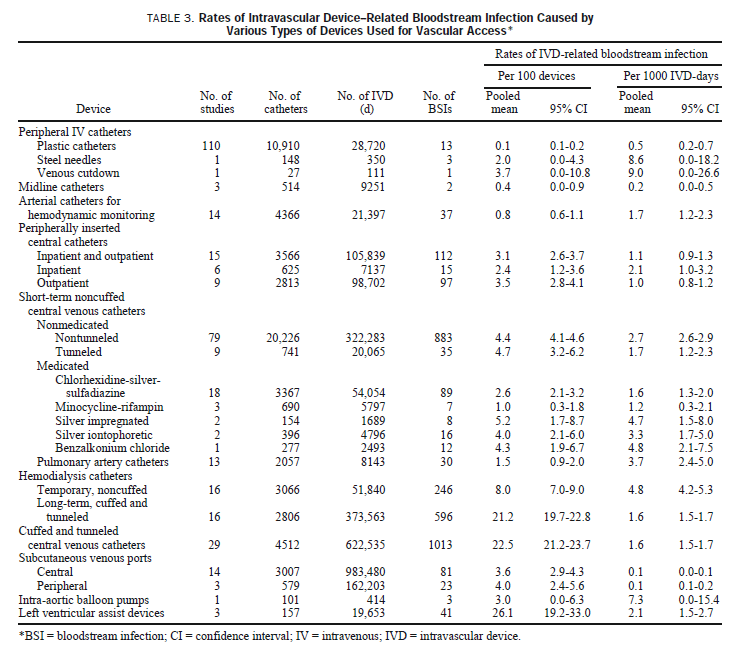
Conclusions: Expressing risk of IVD-related BSI per 1000 IVD-days rather than BSIs per 100 IVDs allows for more meaningful estimates of risk. These data, based on prospective studies In which every IVD in the study cohort was analyzed for evidence of infection by microbiologically based criteria, show that all types of IVDs pose a risk of IVD-related BSI and can be used for benchmarking rates of infection caused by the various types of IVDs In use at the present time. Since almost all the national effort and progress to date to reduce the risk of IVD-related Infection have focused on short-term noncuffed CVCs used in Intensive care units, Infection control programs must now strive to consistently apply essential control measures and preventive technologies with all types of IVDs.
Arvaniti K, et al. Cumulative evidence of randomized controlled and observational studies on catheter-related infection risk of central venous catheter insertion site in ICU patients: a pairwise and network meta-analysis. Crit Care Med. 2017 Apr;45(4):e437-e448. Full-text for Emory users.
Data Synthesis: Twenty studies were included; 11 were observational, seven were randomized controlled trials for other outcomes, and two were randomized controlled trials for sites. We evaluated 18,554 central venous catheters: 9,331 from observational studies, 5,482 from randomized controlled trials for other outcomes, and 3,741 from randomized controlled trials for sites. Colonization risk was higher for internal jugular (relative risk, 2.25 [95% CI, 1.84-2.75]; I2 = 0%) and femoral (relative risk, 2.92 [95% CI, 2.11-4.04]; I2 = 24%), compared with subclavian. Catheter-related bloodstream infection risk was comparable for internal jugular and subclavian, higher for femoral than subclavian (relative risk, 2.44 [95% CI, 1.25-4.75]; I2 = 61%), and lower for internal jugular than femoral (relative risk, 0.55 [95% CI, 0.34-0.89]; I2 = 61%). When observational studies that did not control for baseline characteristics were excluded, catheter-related bloodstream infection risk was comparable between the sites.
Conclusions: In ICU patients, internal jugular and subclavian may, similarly, decrease catheter-related bloodstream infection risk, when compared with femoral. Subclavian could be suggested as the most appropriate site, whenever colonization risk is considered and not, otherwise, contraindicated. Current evidence on catheter-related bloodstream infection femoral risk, compared with the other sites, is inconclusive.
Mermel LA, Allon M, Bouza E, et al. Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2009 Jul 1;49(1):1-45. Erratum in: Clin Infect Dis. 2010 Apr 1;50(7):1079. Dosage error in article text. Erratum in: Clin Infect Dis. 2010 Feb 1;50(3):457. Free full-text.
These updated guidelines replace the previous management guidelines published in 2001. The guidelines are intended for use by health care providers who care for patients who either have these infections or may be at risk for them.
“The density of skin flora at the catheter insertion site is a major risk factor for CRBSI. No single trial has satisfactorily compared infection rates for catheters placed in jugular, subclavian, and femoral veins.” (CDC, p. 27)
CDC (2011, updated 2017). Guidelines for the Prevention of Intravascular Catheter-Related Infections. Atlanta, GA.
More PubMed results on CVC sites and infection rates.
Created 02/12/20; updated 07/20/22.
