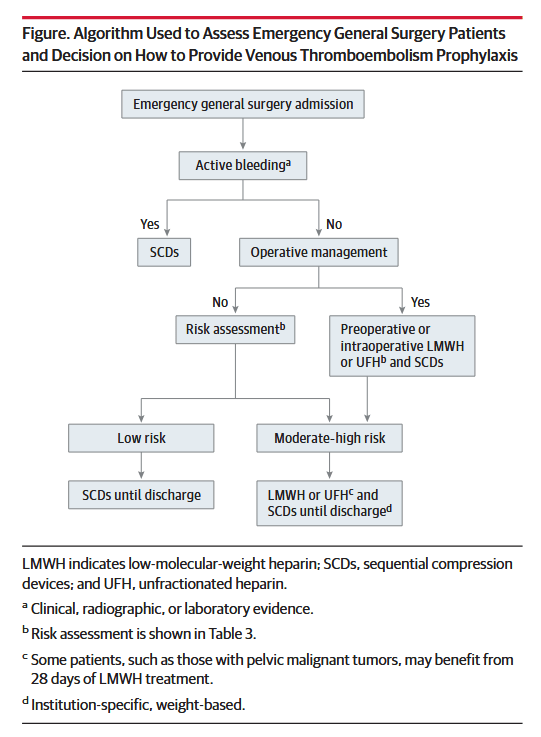
“Venous thromboembolism (VTE) represents the most preventable cause of morbidity and mortality in hospitalized patients, and the Agency for Healthcare Research and Quality (AHRQ) suggests appropriate VTE prophylaxis as a top patient safety practice. The burden of operative and nonoperative emergency general surgery (EGS) is increasing and represents 7% of all hospital admissions in the United States. The reported rate of VTE among patients undergoing EGS is approximately 2.5%. Numerous observational studies, quality improvement studies, randomized clinical trials, reviews, and practice management guidelines are available to guide acute care surgeons in VTE prevention for patients with trauma. However, little guidance is available for the emergency general surgeon. Patients undergoing EGS represent a challenge regarding VTE prevention. Despite the substantial number of annual EGS admissions, little is known about the risk of VTE or the use of mechanical and/or pharmacologic prophylaxis in EGS patients. Furthermore, although guidelines for VTE prophylaxis are available, they are difficult to interpret in the context of admission to an EGS service for an acute condition, particularly when admissions to such services include as many as 70% of patients who do not require operative intervention.”