A discussion during a previous conference included the perioperative management of patients with atrial fibrillation receiving a direct oral anticoagulant (DOAC).
Reference: Douketis JD, et al. Perioperative management of patients with atrial fibrillation receiving a direct oral anticoagulant. JAMA Internal Medicine. 2019 Aug 5; doi:10/1001/jamainternmed.2019.2431
Summary: Each year, 1 in 6 patients with AF, or an estimated 6 million patients worldwide, will require perioperative anticoagulant management. When DOAC regimens became available for clinical use in AF, starting in 2010, no studies had been conducted to inform the timing of perioperative DOAC therapy interruption and resumption, whether heparin bridging should be given, and whether preoperative coagulation function testing was needed. Uncertainty about the perioperative management of DOACs may be associated with unsubstantiated practices and increased harm to patients.
Douketis et al (2019) designed the Perioperative Anticoagulation Use for Surgery Evaluation (PAUSE) study to assess the safety of a standardized perioperative management strategy for a DOAC regimen. They hypothesized that a simple management approach, based on DOAC-specific interruption and resumption intervals, forgoes perioperative heparin bridging, and does not require preoperative coagulation function testing, is safe to use for patient care.
For each DOAC cohort that received DOAC-specific perioperative management, safety was defined as excluding 30-day perioperative rates of major bleeding of 2% and arterial thromboembolism of 1.5%, according to expected outcome rates (1% for major bleeding and 0.5% for arterial thromboembolism) observed with optimal perioperative management of warfarin sodium, and with a proof-of concept prospective study of standardized perioperative dabigatran management. The authors also postulated that this management would yield a high proportion of patients (>90%) with an undetectable or minimal residual anticoagulant level at the time of the procedure.
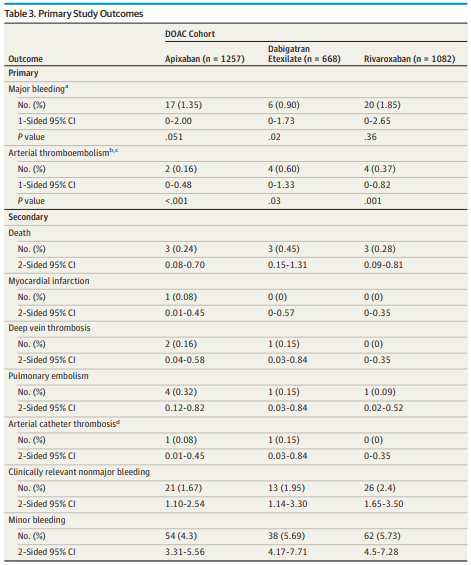
A total of 3640 patients were screened August 1, 2014, through July 31, 2018, from 23 clinical sites in Canada, the United States, and Europe. Of these patients, 3007 (82.6%) were enrolled and included in the primary analysis: 1257 (41.8%) in the apixaban cohort, 668 (22.2%) in the dabigatran cohort, and 1082 (36.0%) in the rivaroxaban cohort. Overall, patients had a mean (SD) age of 72.5 (9.39) years and were predominantly male (1988 [66.1%]).
(p.E7)
Key Findings: In this cohort study of 3007 patients with atrial fibrillation using apixaban, dabigatran, or rivaroxaban, the direct oral anticoagulant treatment was stopped and resumed before and/or after elective surgery or procedure using standardized protocols without heparin bridging. The 30-day postoperative rates of major bleeding were less than 2%, and the rates of stroke were less than 1%.
Key Meaning: In this study, in patients treated with a direct oral anticoagulant, a simple standardized perioperative management approach was associated with low rates of bleeding and stroke.
