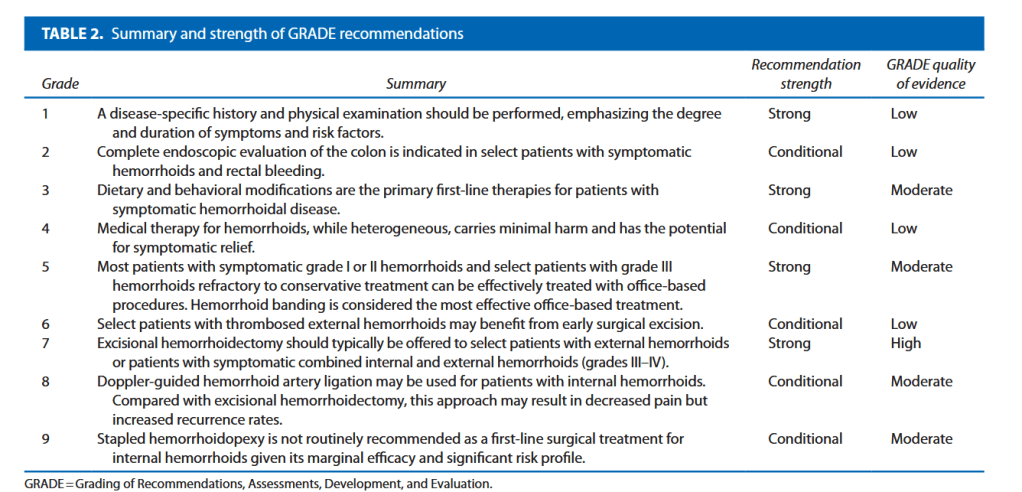
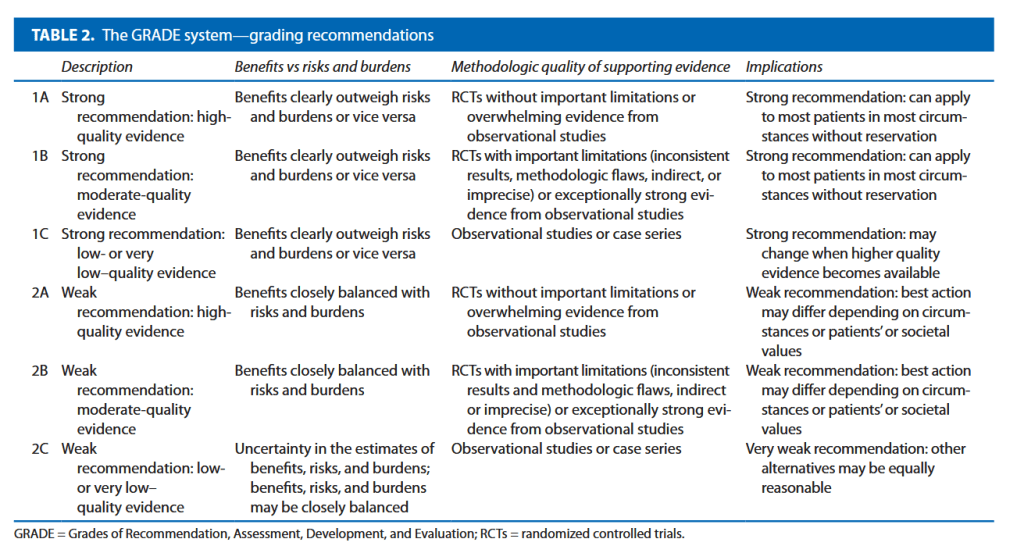
“There is a need for strategies to reduce the risk of ureteric injury, and to facilitate immediate recognition, during colorectal procedures. The preoperative placement of prophylactic ureteric stents or catheters has long been discussed as a technique that may assist colorectal surgeons in identifying and avoiding the ureters, and in recognising ureteric injury when it occurs.
Debate surrounds this topic, however, with no consensus on the precise benefit of prophylactic ureteric stents, and some concerns regarding potential stent-related complications. Whilst the European Association of Urology (EAU) guidelines state that ‘visual identification of the ureters and meticulous dissection in their vicinity are mandatory
to prevent ureteral trauma during abdominal and pelvic surgery’ (grade A recommendation), the use of ‘preoperative prophylactic stents’ are recommended only ‘in selected cases (based on risk factors and surgeon’s experience)’ (grade B). The American Society of Colon and Rectal Surgeons guidelines in surgery for diverticulitis state ‘ureteral stents are used at the discretion of the surgeon’ (grade 2C).
Table 4 Type of repair and outcomes of ureteric injuries
| Study | Injuries (n) | Stented and recognised intraoperatively | Stented and recognised postoperatively | Unstented and recognised intraoperatively | Unstented and recognised postoperatively |
|---|---|---|---|---|---|
| Bothwell [41](open) | 4/561 | 1/4—primary repair over stent | 1/4 (stent insertion had failed)—nephrostomy + stent | 1/4—stent inserted and repair performed | 1/4 re-exploration and ureteroureterostomy |
| Beraldo [32](laparoscopic) | 1/89 | 1/89—repair technique not specified | – | – | – |
| Boyan [34] (laparoscopic) | None | – | – | – | – |
| Chahin [35] | 1/66 | – | 1/66 recognised day 2, managed by retrograde stent reinsertion | – | – |
| Chiu [8] | 2503/811,071 | Not evaluated | – | – | – |
| Coakley [3] | 333/51,125 | Not evaluated | – | – | – |
| Chong [29] | None | – | – | – | – |
| Hassinger [38] | Not evaluated | – | – | – | – |
| Kutiyanawala [44] | 5/251 | No stented patients | No stented patients | 3/5—ureteric re-implant × 2 and ureteroureterostomy over stent in × 1 | 2/5Nephrostomy + JJ stent × 1 (prolonged recovery, fistula)Relaparotomy + removal of ligasure × 1 |
| Kyzer [33] | 1/118 | 1/1 repair technique not specified | – | – | – |
| Leff [19] | 4/194 | 3/4OPEN intraoperative repair | 1/4—delayed presentation as ureteral-cutaneous fistula | Not evaluated | Not evaluated |
| Luks [39] | 2/261 | 2/2 intraoperative repair, type unspecified | – | – | – |
| Merola [28] | 1/374 | None | 1 injury, recognised postoperatively—re-operation (repair not specified) | None | None |
| Nam [27] | None | – | – | – | – |
| Pathak [42] | None | – | – | – | – |
| Palaniappa [45] 2012(Open arm) | 7/4669 | 1/7Ureteroneocystostomy | 1/7Nephrostomy | 3/7Ureteroneocystostomy × 1, ureteroureterostomy × 2 | 2/7Bilateral nephrostomies × 1Ureteroureterostomy × 1 |
| Palaniappa [45]2012(Laparoscopic arm) | 7/1060 | 1/7 Ureteroureterostomy | 1/7 Ureteroneocystostomy | 2/7Ureteroneocystostomy × 1Ureteroureterostomy × 1 | 3/7Ureteroneocystostomy × 1Nephrostomy × 2 |
| Pokala [30] | 0 | – | – | – | – |
| Sahoo [40] | 0 | ||||
| Senagore [36] | 0 | – | – | – | – |
| Sheikh [43] | Not reported | – | – | – | – |
| Speicher [31] | Not reported | – | – | – | – |
| Tsujinaka [37] | 0 | – | – | – | – |