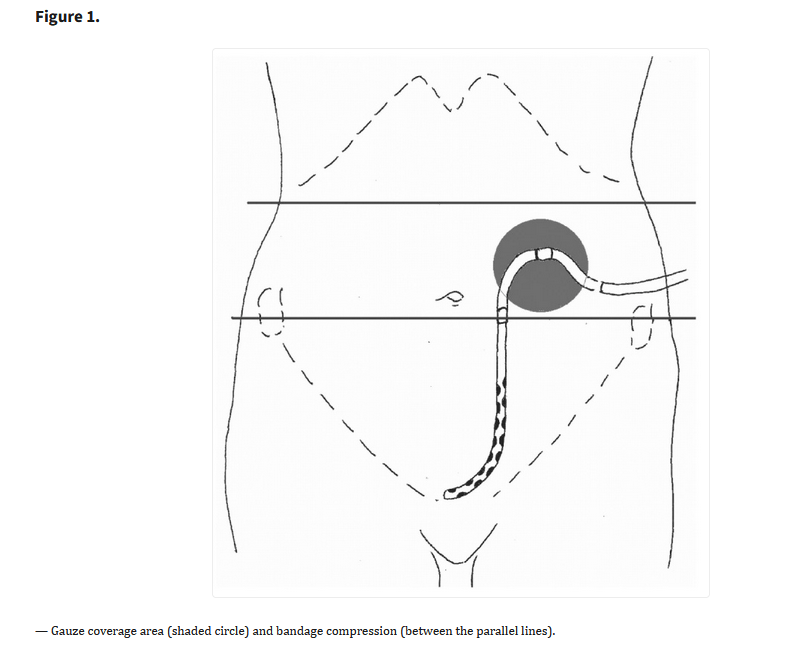
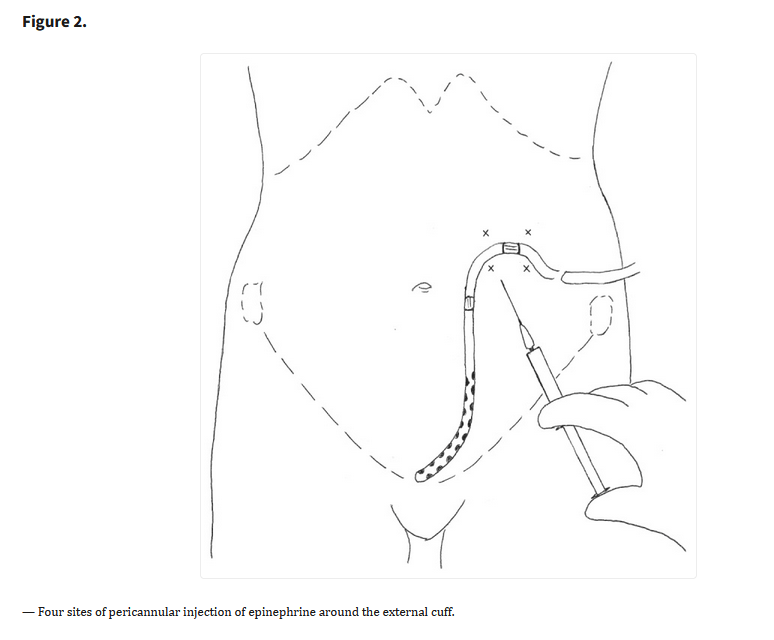
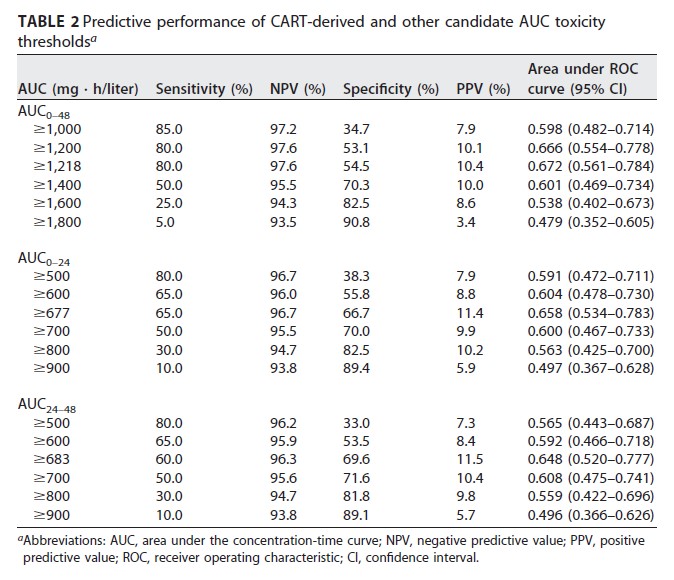
Ferreira GF, et al. Parathyroidectomy after kidney transplantation: short-and long-term impact on renal function. Clinics (Sao Paulo). 2011;66(3):431-5. Free full-text.
Materials and methods: This was a retrospective case-controlled study. Nineteen patients with persistent hyperparathyroidism underwent parathyroidectomy due to hypercalcemia. The control group included 19 patients undergoing various general and urological operations.
Results: In the parathyroidectomy group, a significant increase in serum creatinine from 1.58 to 2.29 mg/dl (P < 0.05) was noted within the first 5 days after parathyroidectomy. In the control group, a statistically insignificant increase in serum creatinine from 1.49 to 1.65 mg/dl occurred over the same time period. The long-term mean serum creatinine level was not statistically different from baseline either in the parathyroidectomy group (final follow-up creatinine = 1.91 mg/dL) or in the non-parathyroidectomy group (final follow-up creatinine = 1.72 mg/dL).
Conclusion: Although renal function deteriorates in the acute period following parathyroidectomy, long-term stabilization occurs, with renal function similar to both preoperative function and to a control group of kidney-transplanted patients who underwent other general surgical operations by the final follow up.
Continue reading →