Cavalcante FP, Millen EC, Zerwes FP, Novita GG. Role of Axillary Surgery After Neoadjuvant Chemotherapy. JCO Glob Oncol. 2020 Feb;6:238-241.
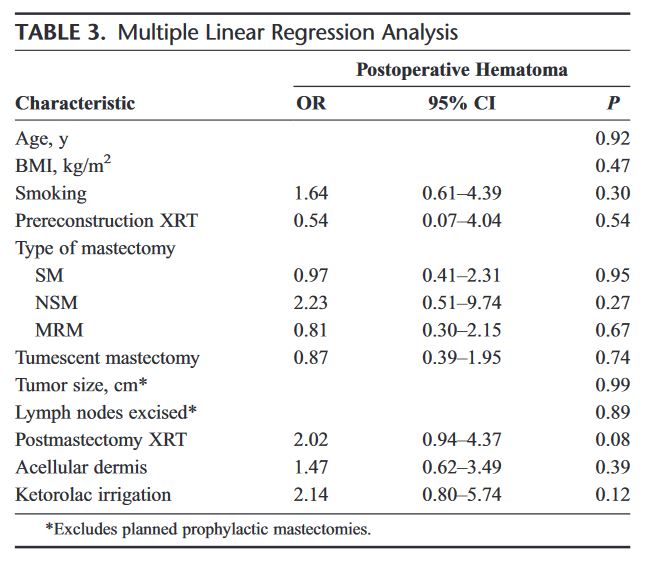
“In a US study, associating selective localization and removal of clipped nodes with SLN dissection, known as targeted axillary dissection, reduced false-negative rates to approximately 2% compared with 4% with removal of the clipped lymph node alone. [20] However, patients are required to undergo two procedures: placement of the clip before systemic treatment and marking it to identify the lymph node during surgery. A retrospective analysis showed that in patients with clipped lymph nodes who were referred for preoperative marking, the clip failed to be identified in 20% of those patients, even when computed tomography was used, with the additional risk of the clip not being removed during surgery. [21] Therefore, the use of clips is controversial, because it is sometimes impossible to remove the clip alone. Despite the association between the number of lymph nodes and false-negative rates, there are still no convincing data regarding clinical outcome.”
Caudle AS, Yang WT, Krishnamurthy S, et al. Improved Axillary Evaluation Following Neoadjuvant Therapy for Patients With Node-Positive Breast Cancer Using Selective Evaluation of Clipped Nodes: Implementation of Targeted Axillary Dissection. J Clin Oncol. 2016 Apr 1;34(10):1072-8.
Results: Of 208 patients enrolled in this study, 191 underwent ALND, with residual disease identified in 120 (63%). The clipped node revealed metastases in 115 patients, resulting in an FNR of 4.2% (95% CI, 1.4 to 9.5) for the clipped node. In patients undergoing SLND and ALND (n = 118), the FNR was 10.1% (95% CI, 4.2 to 19.8), which included seven false-negative events in 69 patients with residual disease. Adding evaluation of the clipped node reduced the FNR to 1.4% (95% CI, 0.03 to 7.3; P = .03). The clipped node was not retrieved as an SLN in 23% (31 of 134) of patients, including six with negative SLNs but metastasis in the clipped node. TAD followed by ALND was performed in 85 patients, with an FNR of 2.0% (1 of 50; 95% CI, 0.05 to 10.7).
Conclusion: Marking nodes with biopsy-confirmed metastatic disease allows for selective removal and improves pathologic evaluation for residual nodal disease after chemotherapy.
Continue reading →