A discussion this week included signet ring cell carcinoma.
Reference: Nitsche U, et al. Mucinous and signet-ring cell colorectal cancers differ from classical adenocarcinomas in tumor biology and prognosis. Annals of Surgery. 2013 Nov;258(5):775-782; discussion 782-783. doi:10.1097/SLA.0b013e3182a69f7e
Additional Reading: Korphaisarn K, et al. Signet ring cell colorectal cancer: genomic insights into a rare subpopulation of colorectal adenocarcinoma. British Journal of Cancer. 2019 Sep;121(6):505-510. doi:10.1038/s41416-019-0548-9
Summary: In a study analyzing clinical, histopathological, and survival data of 3479 patients undergoing surgery for primary colorectal cancer between 1982 and 2012, Nitsche et al (2013) compared the characteristics of classical adenocarcinomas (AC) to the less common mucinous adenocarcinomas (MAC) and to the rare signet-ring cell carcinomas (SC).
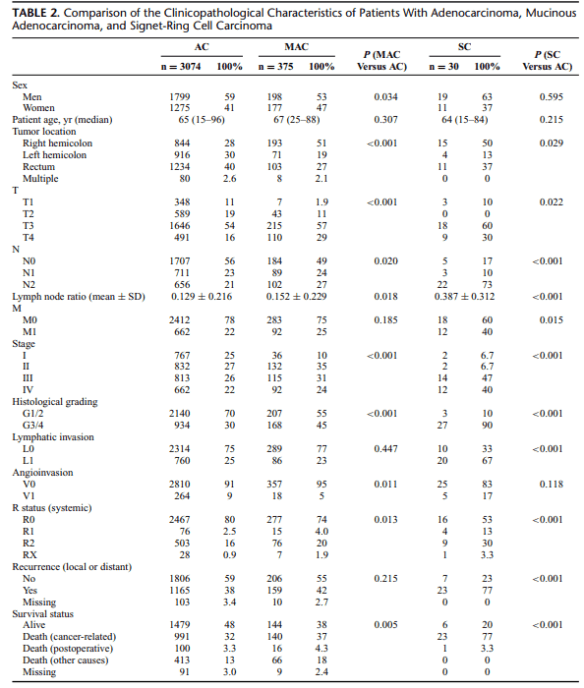
Approximately 10% of all colorectal cancers are MAC, and about 1% are SC. Because of their relatively rare occurrence, in particular, the evaluation of the clinical impact of SC is difficult. However, compared with AC, both MAC and SC have been shown to be associated with young age, advanced tumor stage, accumulation in female patients, and distinct molecular patterns, such as microsatellite instability and activating mutations of the BRAF gene. Although ambiguous, recent data and meta-analyses suggest that the
histological subtype MAC may be associated with worse outcome compared with AC. Poor prognosis of SC is more evident, mainly due to high rates of synchronous and metachronous distant organ metastasis associated with this histological subtype.
In describing SC, the authors state: “SC have been described as being positive for intestinal trefoil factor and MUC2, 2 peptides that are usually produced only by goblet cells. Thus, SC could arise from different cells of origin than AC. Although they can be localized in the colorectum, SC may be genetically more related to signet-ring cell cancers of other organs (eg, gastric cancer) than to AC or MAC of the colorectum. The
absence of E-cadherin/β-catenin and amplification of Bcl-2 are features typically shared with signet-ring cell cancer of the stomach but not with classical colorectal adenocarcinomas” (p.781).
The authors conclude that patients with MAC and SC could profit from closer follow-up or even intensified adjuvant therapy because of their high rates of local and distant recurrence. The biological behavior of SC differs in specific, and these patients require special awareness, despite the relatively rare prevalence.



 (Cerek et al, 2018)
(Cerek et al, 2018)