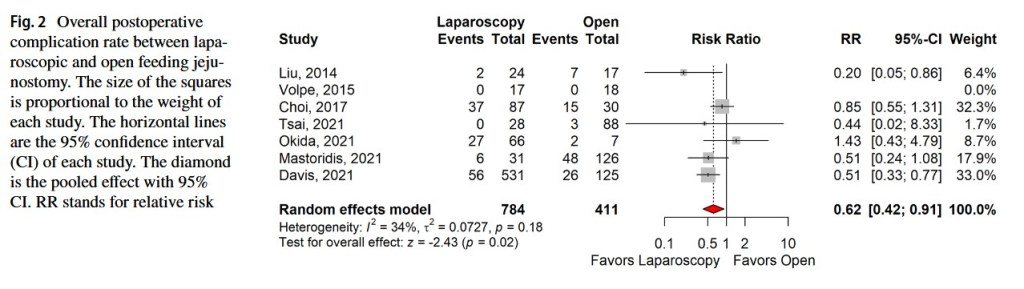
“Feeding jejunostomy is conventionally done via laparotomy. However, since laparoscopic jejunostomy was introduced in 1990, the procedure is constantly advancing with new techniques and devices. It was demonstrated to be a safe, feasible, and cost-effective technique with comparable complication rate to open feeding jejunostomy.”
“In addition, compared with the conventional open procedure, the laparoscopic approach has the inherit merits of smaller incisions, better cosmesis, less postoperative pain, and earlier recovery. With the aim of achieving early enteral feeding and a reduction in postoperative morbidity, any complications arising from the procedure will jeopardize its benefits, incur additional costs, and delay subsequent oncologic treatment.”