“Chronic pancreatitis is characterized by recurrent inflammation and fibrosis, resulting in pervasive symptoms of abdominal pain, early satiety, nausea, malnutrition, and pancreatic insufficiency. Though there are limited data on the true prevalence of chronic pancreatitis, an
estimated 5 to 14 per 100,000 patients are diagnosed annually in the US. While the overall incidence and prevalence of chronic pancreatitis remain relatively low, it contributes a significant morbidity and financial burden, with an annual healthcare cost exceeding $3 billion, largely due to increased utilization and symptom palliating efforts. Furthermore,
disability secondary to chronic pancreatitis symptoms creates a substantial personal burden, with increased work absenteeism and reducedquality of life. Treatment efforts initially focus on symptom management and reversal of instigating factors, consisting primarily of medical
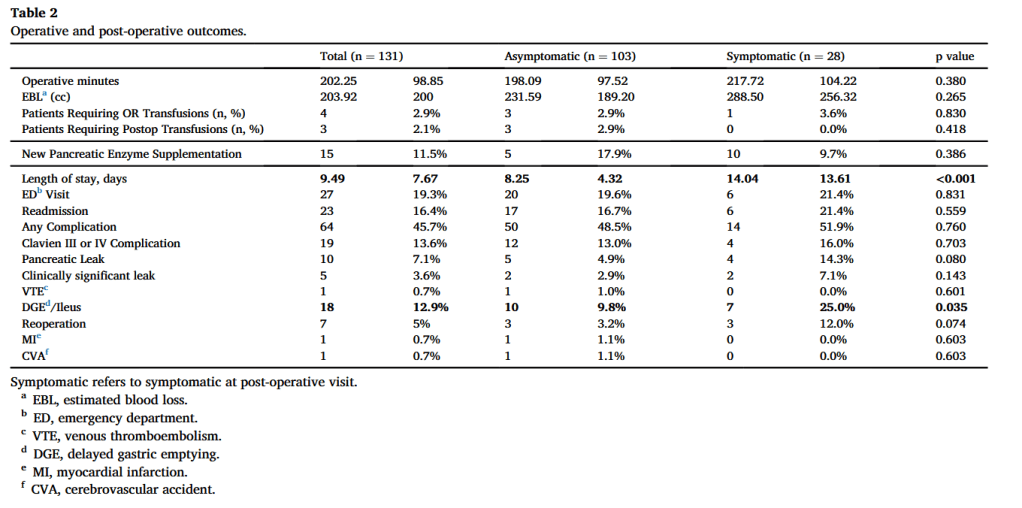
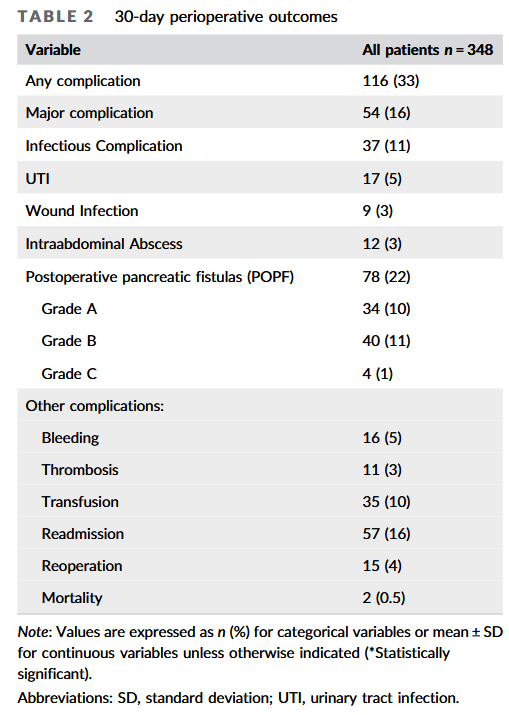
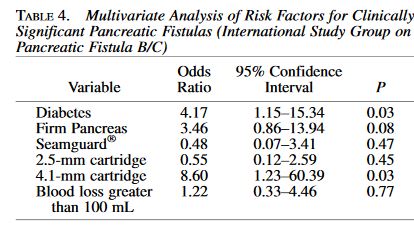
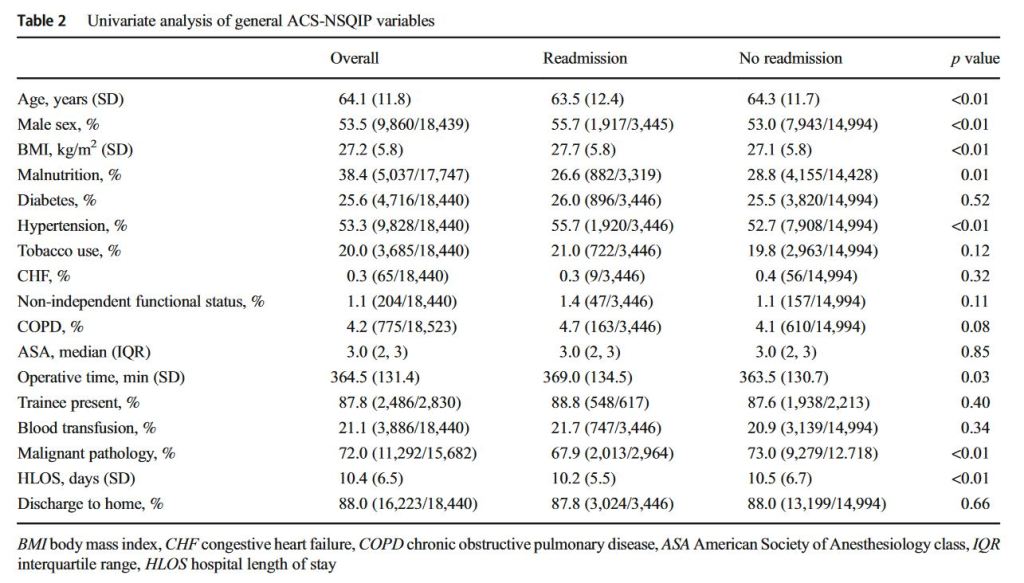
and endoscopic techniques; however, up to 50 % of all cases of chronic pancreatitis eventually require surgical intervention due to persistent symptoms, most commonly debilitating abdominal pain. Additionally, current data suggest that surgery is superior to endoscopy in maintaining symptom resolution and preserving pancreatic function.”