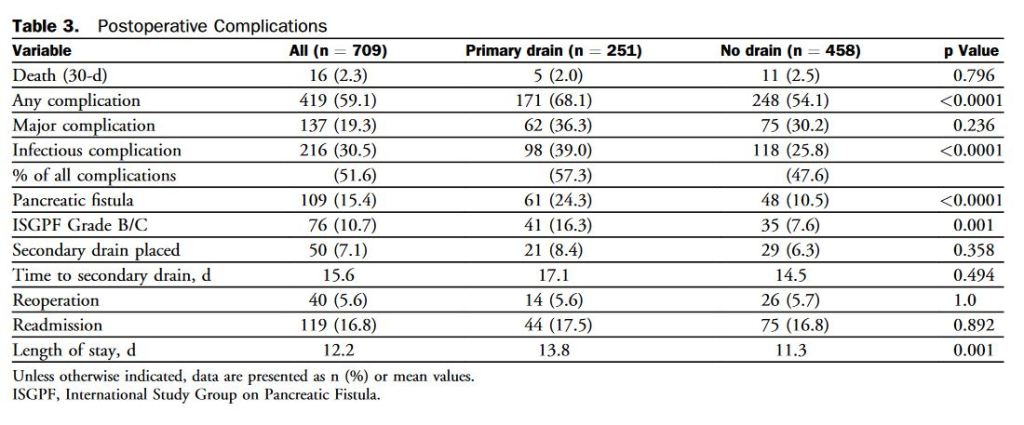
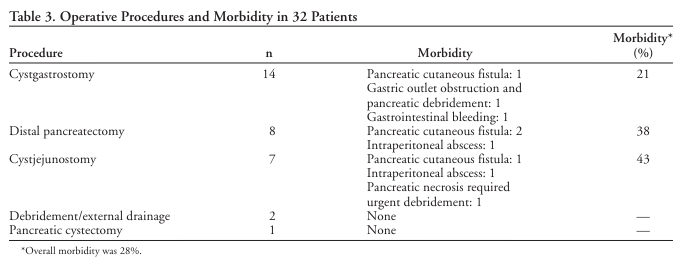
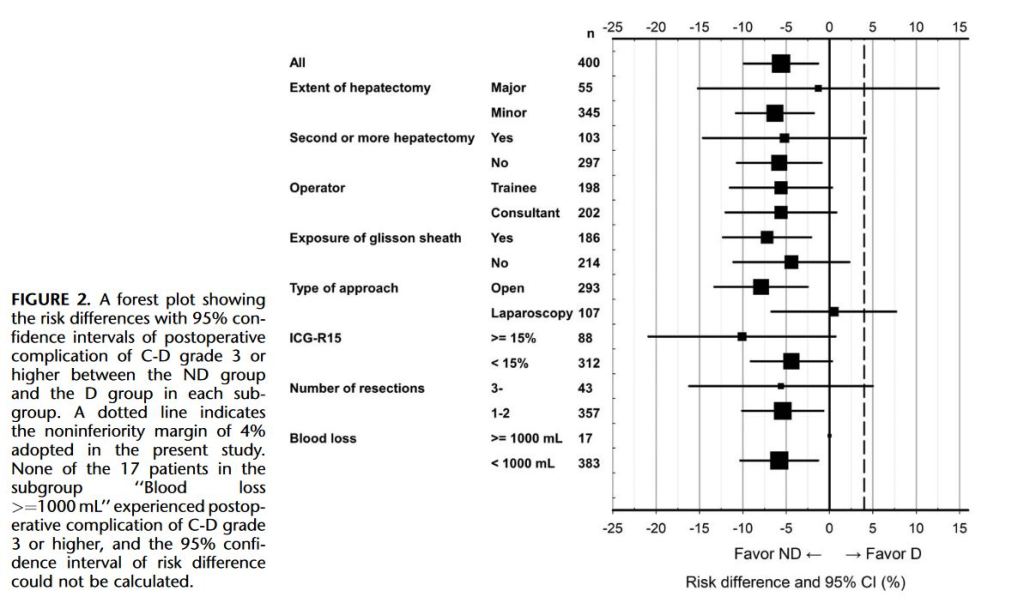
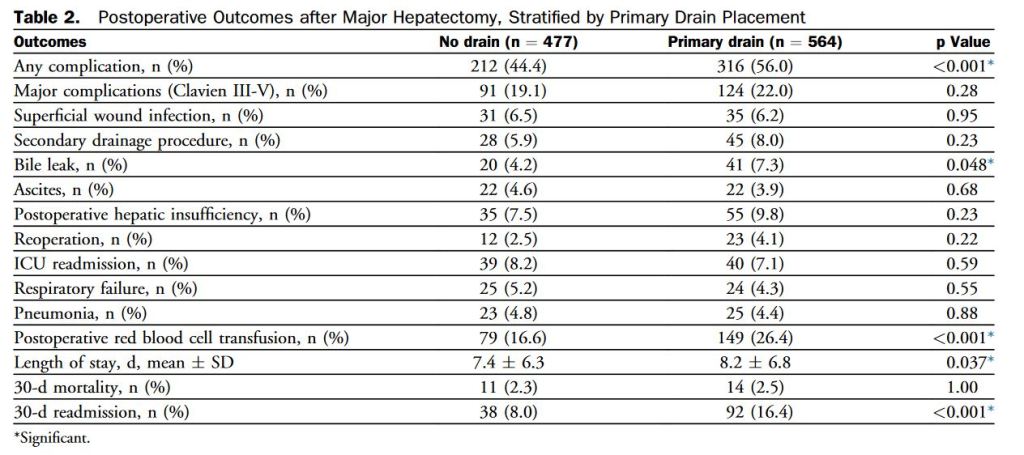
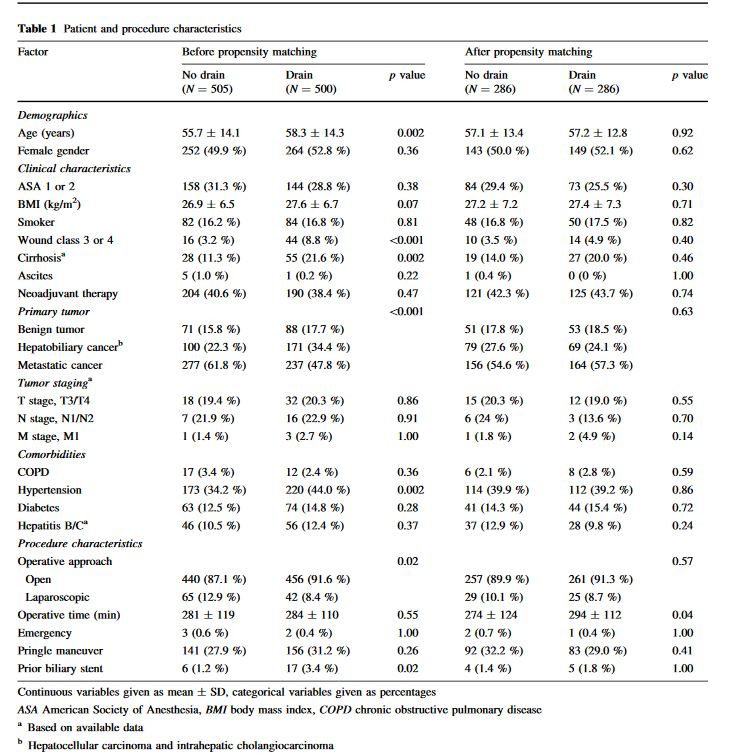
“Postoperative pancreatic fistula (POPF) is a potentially devastating complication after pancreatic resection, seen in 5% to 30% of patients. Depending on severity, POPF may be associated with infectious complications, reoperation, increased length of hospital stay, readmission, and even death. Historically, surgeons placed drains routinely for many abdominal procedures to control potential leaks from various anastomoses. However, this practice has been abandoned by many surgeons over the last 2 decades because no benefit to routine abdominal drainage has been observed from several randomized controlled trials for resections of the colon and rectum, gallbladder, and liver.”